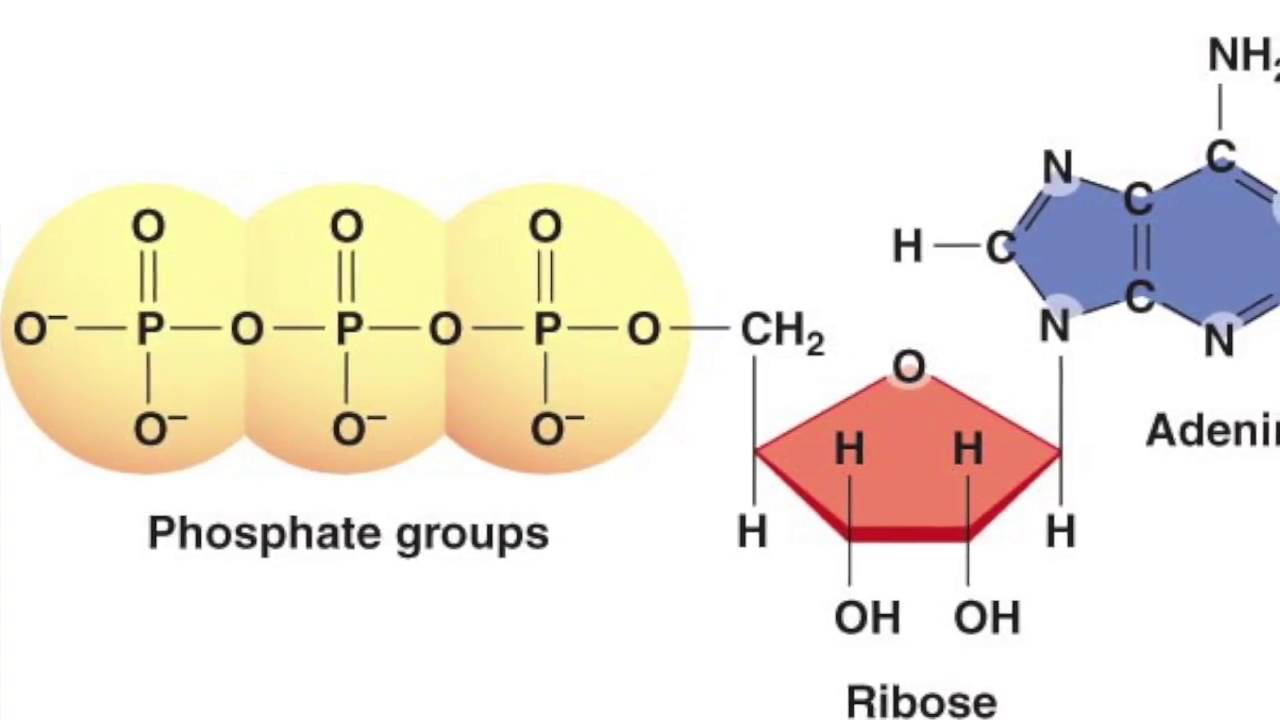
In many such cases the β phosphoanhydride bond is cleaved, generating pyrophosphate. In this case the reaction may involve little of no free energy change, so that it would appear to be easily reversible. In certain reactions of ATP the energy of a phosphoanhydride bond is being used to created a bond of similar free energy of hydrolysis, which can itself be used subsequently to activate other reactions. ribose-3′OH + ATP → -ribose-3′O-PO 2-O-5′-Adenosine + PPiĪTP and ‘activation’ reactions: rationale for pyrophosphate The obvious example is the RNA polymerase reaction, which for our purposes can be represented as: In the second type of reaction the AMP component of ATP (or similar molecules) is incorporated into one of the products, so that the other product must be pyrophosphate. The reaction is essentially irreversible as the energy of the hexose phosphate bond is much less than that of the phosphoanhydride bond. A simple example of this is the hexokinase reaction: In the first type of reaction a phosphate group is incorporated into the product, in which case this will obviously be the γ-phosphate and ADP will be generated. In this case the actual chemical reaction determines which bond is hydrolysed. In some cases there is not only hydrolysis of a phosphoanhydride bond, but a chemical component of ATP is incorporated into one of the products. (Hydrolysis of the β bond would ‘loose’ the second bond in pyrophosphate, where its hydrolysis cannot be used productively.) An example is the conversion of pyruvate to oxaloacetate in gluconeogenesis, in which energy is required to form a carbon–carbon bond:ĬH 3COCOO – + HCO 3 – + ATP → COO –CH 2COCOO – + ADP + Pi The energy of hydrolysis of only a single phosphoanhydride bond is sufficient for this, and is achieved by the hydrolysis of the γ bond with the production of ADP. In this hypothetical example, 20 kJ/mol of the free energy of hydrolysis of ATP is used to convert A to B, the other 10 kJ/mol is lost as heat, but makes the reaction essentially irreversible. In many cases the hydrolysis of ATP is coupled to an energetically unfavourable reaction (one with a +ve ΔG o) so that the coupled reaction has overall negative free energy change and hence the energetically favourable reaction can take place: A → B ΔG = 20 kJ/mol (unfavourable)ĪTP → ADP + Pi ΔG = –30 kJ/mol (favourable)Ī + ATP → B + ADP + Pi ΔG = –10 kJ/mol (favourable) Use of ATP to drive unfavourable reactions The hydrolysis of the β- and γ- phosphoanhydride bonds of ATP both have a high negative values of ΔG o of ca. The actual free energy change, ΔG, depends on the concentration of reactants and products (mass action effects). the values obtained under conditions in which the concentration of all reactants and products are set at the same concentration. The literature provides values of the standard free energy changes, ΔG o, for reactions, i.e. An input ofįree energy is required to drive such a reaction. A reaction cannot occur spontaneously if ΔG is positive.A system is at equilibrium and no net change can take place if ΔG is zero.A reaction can occur spontaneously only if ΔG is negative.This is because, to quote from Berg et al.: In considering biological reactions, it is usual to adopt a thermodynamic approach in which the thermodynamical (Gibbs) Free Energy change (ΔG) of the reaction is considered. I have discussed some of this at greater length in an answer to another question, however it is important to clarify this at the outset. If the free-energy of hydrolysis of the phosphoanhydride bond is being ‘transferred’ to another molecule in order to ‘activate’ it for a specialized group-transfer role (often in the synthesis of macromolecules) two bonds are often broken: the first (β-) transfers the free energy with the production of pyrophosphate, whereas the second (now in the pyrophosphate) is hydrolysed non-productively (free energy lost as heat) to ensure that the overall reaction is irreversible.Įnergy considerations in reactions involving ATP If the hydrolysis (with a negative free energy change, ΔG) is being coupled to a reaction with a positive ΔG, hydrolysis of single (γ-) phosphoanhydride bond usually suffices, with the production of ADP and Pi. If a part of the ATP molecule is incorporated into one of the products, the choice of bond to be cleaved emerges from the chemistry of the reaction. It is the nature of the biochemical reaction that determines whether a reaction of ATP involves hydrolysis of the β- or γ- phosphoanhydride bond.
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
